
Some of the important properties of the cathode rays studied by Sir J.J. 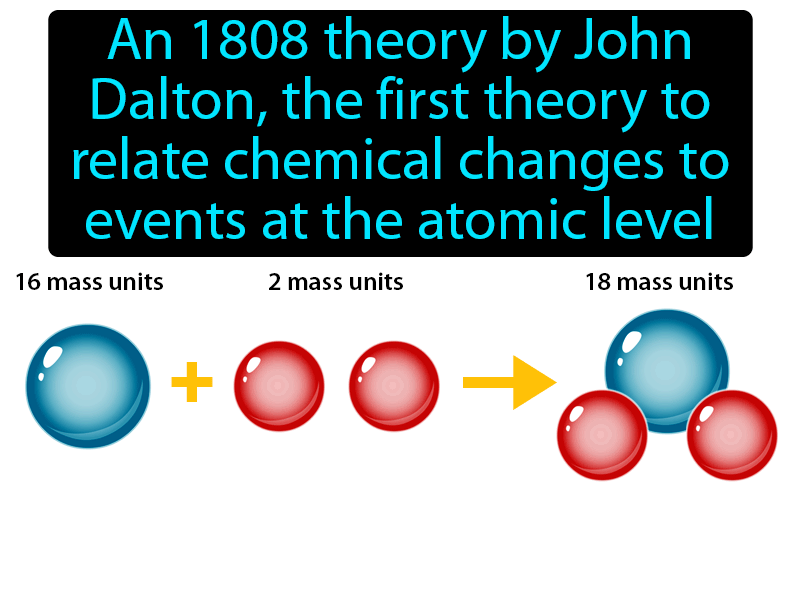
When a current of high voltage (10,000 volts) is passed through a gas of air kept at a very low pressure (0.01 – 0.03 mm) blue rays are seen emerging from the case. The passage of electricity through gases as studied by a number of physicists, particularly by Faraday, Davy, Crookes and J.J. Though Dalton’s atomic theory could not give a convincing explanations to any of the above facts, it laid the foundation for the development of Modern atomic theory.The basic postulate of Dalton’s atomic theory which says that “Atom’s are the tiniest particles of matter which take part in Chemical reaction” is however accepted in modern atomic theory with experimental evidence.ĭuring the latter half of the nineteenth century, it was found that while normally dry gases do not conduct an electric current, they do so under very low pressure and then patches of light are seen. atoms of same elements may not be identical in all respect. the electron, proton, and neutron, it was concluded that atom can be further divided.ĭiscovery of isotopes proved that atoms of the same element may possess different atomic weights. With the discovery of subatomic particles, i.e. At present about 35 different subatomic particles are known but the three particles namely electron, proton and neutron are regarded as the fundamental particles.ĭalton’s atomic theory has been contradicted with the advancement of science and modified on the basis of further research and discoveries as follows: 
However, the researches done by various eminent scientists and the discovery of radioactivity have established beyond doubt that atom was not the smallest indivisible particle but has a complex structure of its own and was made up of still smaller particles like electrons, protons, neutrons etc. This means that a chemical reaction is just a simple rearrangement of atoms and the same number of atoms must be present before and after the reaction.Ītoms of different elements combine in a fixed ratio of small whole numbers to form compound atoms called molecules. Matter is made up of small indivisible particles called atom.Ītom is the smallest particle of an element, which takes part in a chemical reaction.Ītoms of the same element are identical in all respects especially in size, shape and mass.Ītoms of different elements have different mass, shape and size.Ītoms can neither be created nor be destroyed. The main postulates of Dalton’s atomic theory are: However, he made a mistake in assuming that the simplest compound of two elements must be binary, formed from atoms of each element in a 1:1 ratio, and his system of atomic weights was not very accurate - he gave oxygen an atomic weight of seven instead of eight.ĭespite these errors, Dalton's theory provided a logical explanation of concepts, and led the way into new fields of experimentation.On the basis of large number of experiments performed, John Dalton proposed that matter was made up of extremely small particles called atoms. Using his theory, Dalton rationalised the various laws of chemical combination which were in existence at that time. The theory proposed a number of basic ideas:Īll atoms of the same element are identicalĭifferent elements have different types of atomsĬhemical reactions occur when atoms are rearrangedĬompounds are formed from atoms of the constituent elements. He stated his theory in a lecture to the Royal Institution in 1803. Dalton's theory was based on the premise that the atoms of different elements could be distinguished by differences in their weights.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
